An AMPKa2-specific phospho-switch controls lysosomal targeting for activation
Journal article
Morrison, Kaitlin R., Smiles, William J., Ling, Naomi X. Y., Hoque, Ashfaqul, Shea, Gabrielle, Ngoei, Kevin R. W., Yu, Dingyi, Murray-Segal, Lisa, Scott, John W., Galic, Sandra, Kemp, Bruce E., Petersen, Janni and Oakhill, Jonathan S.. (2022). An AMPKa2-specific phospho-switch controls lysosomal targeting for activation. Cell Reports. 38(7), p. Article 110365. https://doi.org/10.1016/j.celrep.2022.110365
| Authors | Morrison, Kaitlin R., Smiles, William J., Ling, Naomi X. Y., Hoque, Ashfaqul, Shea, Gabrielle, Ngoei, Kevin R. W., Yu, Dingyi, Murray-Segal, Lisa, Scott, John W., Galic, Sandra, Kemp, Bruce E., Petersen, Janni and Oakhill, Jonathan S. |
|---|---|
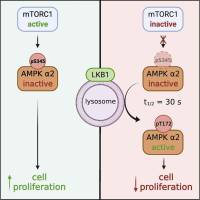
| Abstract | AMP-activated protein kinase (AMPK) and mechanistic target of rapamycin complex 1 (mTORC1) are metabolic kinases that co-ordinate nutrient supply with cell growth. AMPK negatively regulates mTORC1, and mTORC1 reciprocally phosphorylates S345/7 in both AMPK α-isoforms. We report that genetic or torin1-induced loss of α2-S345 phosphorylation relieves suppression of AMPK signaling; however, the regulatory effect does not translate to α1-S347 in HEK293T or MEF cells. Dephosphorylation of α2-S345, but not α1-S347, transiently targets AMPK to lysosomes, a cellular site for activation by LKB1. By mass spectrometry, we find that α2-S345 is basally phosphorylated at 2.5-fold higher stoichiometry than α1-S347 in HEK293T cells and, unlike α1, phosphorylation is partially retained after prolonged mTORC1 inhibition. Loss of α2-S345 phosphorylation in endogenous AMPK fails to sustain growth of MEFs under amino acid starvation conditions. These findings uncover an α2-specific mechanism by which AMPK can be activated at lysosomes in the absence of changes in cellular energy. |
| Keywords | kinase; metabolic signaling; phosphorylation; AMPK; mTORC1; lysosome; energy homeostasis |
| Year | 2022 |
| Journal | Cell Reports |
| Journal citation | 38 (7), p. Article 110365 |
| Publisher | Cell Press |
| ISSN | 2211-1247 |
| Digital Object Identifier (DOI) | https://doi.org/10.1016/j.celrep.2022.110365 |
| PubMed ID | 35172150 |
| Scopus EID | 2-s2.0-85124564701 |
| Open access | Published as ‘gold’ (paid) open access |
| Page range | 1-13 |
| Funder | National Health and Medical Research Council (NHMRC) |
| Australian Research Council (ARC) | |
| Flinders Foundation | |
| Flinders University | |
| St Vincent’s Institute of Medical Research | |
| Operational Infrastructure Support (OIS) Program, Victorian Government | |
| Publisher's version | License File Access Level Open |
| Output status | Published |
| Publication dates | |
| Online | 15 Feb 2022 |
| Publication process dates | |
| Accepted | 19 Jan 2022 |
| Deposited | 27 Oct 2023 |
| Supplemental file | License File Access Level Open |
| ARC Funded Research | This output has been funded, wholly or partially, under the Australian Research Council Act 2001 |
| Grant ID | GNT1145836 |
| GNT1138102 | |
| GNT1161262 | |
| GNT1161262 | |
| DP180101682 |
https://acuresearchbank.acu.edu.au/item/8zx8v/an-ampka2-specific-phospho-switch-controls-lysosomal-targeting-for-activation
Download files
Publisher's version
| OA_Morrison_2022_An_AMPKa2_specific_phospho_switch_controls.pdf | |
| License: CC BY 4.0 | |
| File access level: Open | |
Supplemental file
| OA_Morrison_2022_An_AMPKa2_specific_phospho_switch_controls_[GRAPHICAL_ABSTRACT].jpg | |
| License: CC BY 4.0 | |
| File access level: Open | |
122
total views130
total downloads5
views this month4
downloads this month